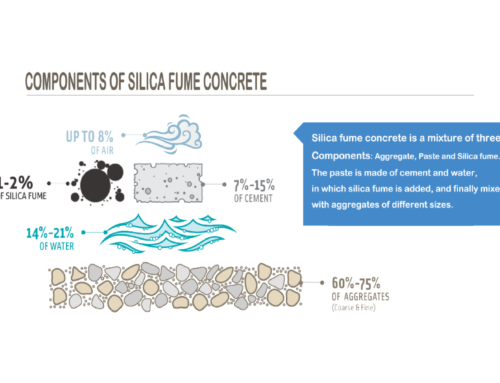
This article examines how silica fume changes the properties of paste, mortar, and concrete. The effects of silica fume are attributed to physical and chemical mechanisms, both of which are described. Because the results of both the physical and chemical mechanisms are seen in modifications to the paste microstructure, Additionally, topics that are related to the mechanism of silica fume reactions, such as autogenous shrinkage, pore water chemistry, reactions with different types of portland cement, the heat of hydration, and reactions with chemical admixtures.
silica fume(15%) replacement of cement, there are approximately 2 million particles of silica fume for each grain of portland cement in a concrete mixture. Therefore, it is no surprise that silica fume has a pronounced effect on concrete properties. All of the physical and chemical mechanisms described depending on the thorough dispersion of the silica-fume particles to be effective. This requires the addition of sufficient quantities of WRAs to overcome the effects of surface forces, ensure good dispersion, and thereby promote good packing of the solid particles. The addition of materials to the mixer in the proper sequence, as well as thorough mixing, are also essential.
Reduced bleeding
The presence of silica fume in fresh concrete results in significantly reduced bleeding and greater cohesiveness. This is a physical effect, the result of incorporating extremely fine particles into the mixture. The higher the silica fume content, the more pronounced the effect.
The increased coherence (cohesiveness) will benefit the hardened concrete structure in terms of reduced segregation and bleed water pockets under reinforcing bars and coarse aggregate.
Provision of nucleation sites
Silica fume accelerates the hydration of cement during the early stages by providing nucleation sites where the products of cement hydration can more readily precipitate from solution.
Particle packing
In hardened concrete, silica-fume particles improve the packing of the solid materials by occupying some of the spaces between the cement grains in the same way as cement occupies some of the spaces between the fine-aggregate particles, and fine aggregate
occupies some of the spaces between coarse-aggregate particles in concrete. This analogy applies only when the surface forces between the cement particles are negligible, that is, when there is enough WRA present to overcome the effects of surface forces.
Chemical effects
Silica fume is a highly reactive pozzolan. In hydrating cement paste, silica fume will react with CH to form calcium-silicate hydrate (CSH).
silica fume contacts mixing water and forms a silica-rich gel, absorbing most of the available water. This gel then agglomerates between the grains of unhydrated cement, coating the grains in the process. CH reacts with the outer surface of this gel to form CSH. This silica-fume gel CSH forms in the voids of the CSH produced by cement hydration, thus producing a very dense structure.